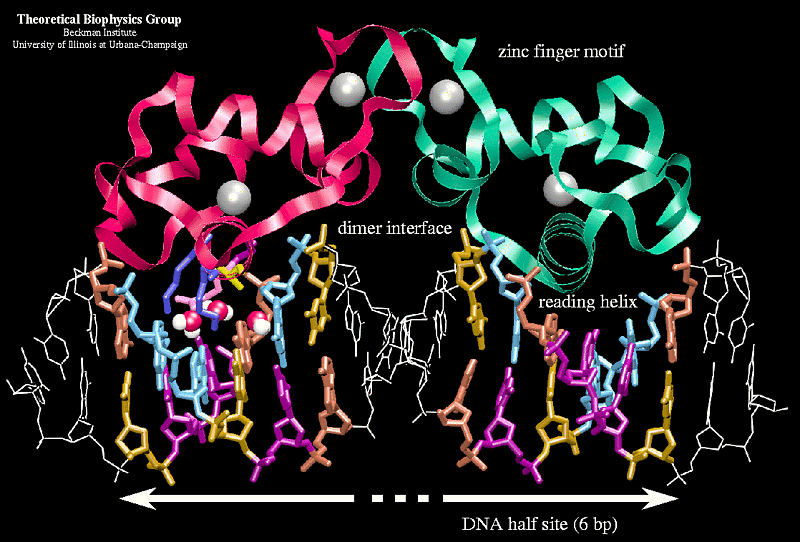16 Nuclear Receptors|Tulane

Synchronizing cells for cell cycle studies
• Drugs – Aphidicolin, hydroxyurea, nocodazole, serum starvation, mimosine
• Mitotic shake-off
• Centrifugal elutriation
• Embryonic cells
• Yeast
Yeast
The yeast mitotic cycle
Yeast must respond to size, food and sex-START. Somatic cells must grow
and reach a certain size before division.
- yeast cycles in 90 minutes.
- Grows in simple medium.
- Well-defined genetics.
- Size of bud or length defines cell cycle position.
Schizosaccharomyces pombe – fission yeast
Saccharomyces cerevisiae – budding yeast.
The G2 checkpoint is more prominent in pombe.
The fission yeast S. pombe.
https://www.youtube.com/watch?v=nhMFxqC89Ac
Wild-type cell division cycle (CDC) genes can be isolated from a genomic library by functional complementation of cdc mutants
Paul Nurse isolated cdc2 using complementation assays in S. Pombe Also cdc13
- Cdc2 sequence reveals a protein kinase.
- Antibody to cdc2 immunoprecipitates MPF.
- Cdc13 has homology to cyclin B.
- MPF is a heterodimer of cdc2/cdc13 with protein kinase activity.
ts mutant phenotypes: Wee1 sequence reveals a protein kinase, Cdc25 is a protein phosphatase
- Change in the amount of cdc2 or a change in the activity? Gene dosage in wild-type cells
| Genotype | Size at mitosis |
|---|---|
| 5 x cdc2 | no effect |
| 5 x cdc13 | no effect |
| Δwee1 | small |
| 3 x wee1 | large |
| 5 x cdc25 | small |
Cdc2: Complex with Cycline B, phosphate on Y161. Activating
Y14, deactivate
CDK1 – human gene complements cdc2 mutant. 65% identical to pombe cdc2. All eucaryotes have a cdc2 gene. MPF required for mitosis – works in the absence of protein synthesis.
Gene dosage experiments demonstrate that extra copies of cdc13 don’t control M.
- How is the activity of MPF controlled?
Human cdc2/CDK1 Y-15 phosphorylation inhibits and T-161
phosphorylation activates.
Also requires cyclin binding.
Targets for MPF?
• nuclear lamins A&C (alternative splice products of a single gene) lamin B stays associated with nuclear membrane through isoprenylation.
• SMC (structural maintenance of chromosomes) proteins, which form condensin, a multi-protein complex. Addition of an anti- SMC antibody to xenopus extracts prevents DNA condensation and association of sister chromatids.
• SMC must be phosphorylated to wind DNA into supercoils. SMC is also a component of cohesin, which holds sister chromatids together at the centromere. Another component of cohesin is Scc1, a target for separase.
The nuclear lamina and its regulation by phosphorylation
Polo and aurora kinases Remove cohesins from arms
Separation of chromosomes
Cdk-cyclin B phosphorylation promotes APC/C-cdc20 interaction. In the transition from metaphase to anaphase the Scc1 (sister chromatid cohesion protein1) subunit of cohesin is degraded by separase, a caspase-like protease. Ubiquitination of securin, a separase inhibitor, is directed by cdc20-APC-starts onset of anaphase.
Attachment of last kinetochore activates securin destruction.
Exiting Mitosis
Ubiquitin linked to cysteine on E2 transferred to lysine of target
- MPF must be inactivated for cells to exit mitosis.
- MPF activates ubiquitin conjugation of cyclin B. 9 a.a. destruction box.
- Cyclin B must be cdc2 associated to be degraded.
- Anaphase Promoting Complex (APC) activated by MPF-promotes APC-cdc20 interaction. Inactivated by G1 cyclin-CDK. Required for entry into anaphase and exit into G1
APC substrates stable in S, G2 and M. Unstable metaphase to anaphase and in G1. APC orders mitosis. How?
Regulation of S phase and mitotic cyclin levels
Cdh1-APC degrades cyclin to exit anaphase. Cdh1 inhibited by MPF. Describe Cdk1 activity during the phases of mitosis.
Cdh1 is hct1 in pombe. After chromosomes segregate cdc14 is activated –released from sequestration by a GTP-binding protein.
Cdc14 also induces transcription of SIC1.
The embryonic cell cycle of xenopus laevis
Cleavage without growth- 12 consecutive mitoses – no RNA synthesis
Oocyte stockpiles materials for multicellular embryo
What signal does cdc14 respond to? Chromosome separation in anaphase activates cdc14. Cdc14 is a phosphatase that targets cdh1
Ordering Mitosis
APC/C 13 subunits in yeast.
CyclinA-GFP fusion protein demonstrates it is the first substrate of APC/C. If cyclin A can’t be degraded, unable to initiate anaphase.
APC/C-cdc20 ubiquitinates securin leading to its degradation and activation of separase which degrades Scc1 in the transition from metaphase to anaphase.
Cdc14 activates APC/C-cdh1 to degrade cyclin
Cdc20 is a substrate for APC/C-cdh1. How does this affect activity?
Mitotick Checkpoint
Cdc28 vs cdc2
Sic→↑↑↓
17 let’s see what cat we record
Page1
Markers of the CDK kinase domain
Page2:
Classify:
A type cycline, D type cycline
Page3:
Predicted Cyclin-binding elements
Page4:
Cdk 7 and 8 involved in transcription
Page5:
Elaborate explained table
Page6:
Graph:
mammalian cell, gg roal?
CyclinD starte close serum
Discrib the S phase by the information from previous table.
CDK2 + Cyclin E and A resbonsible to the entry of the S phase.
P7:
||Vertebrates |Budding yeast|
| Cyclin-cdk complex | cyclin | cdk | Cyclin | cdk |
|---|---|---|---|---|
| G1-cdk | D | Cdk4 & cdk6 | Cln3 | cdc-28 |
| G1/S cdk | E | Cdk2 | Cln1 & cln2 | cdc-28 |
| S-cdk | A | Cdk2 | Clb 5 & clb 6 | cdc-28 |
| M-cdk | B | Cdk1 | Clb1,2,3,4 | cdc-28 |
P8:
CAK → CDK7 → tf2b tf2h
TF2A form complex to
Wee1 Kinase → Inhibits CAK
Cdc25 phosphatase
Cdc25A (Virtbrates)
Cdc25C (Virtbrates)
ATM/ATR kinase
Chk1/Chk2 kinase
P15:
IRES: internal ribosome entry site
Oncogenic Viruses
Viruses
- Simple obligate intracellular parasites of plants, animals, and bacteria.
- Target to the cell surface receptor.
- Consist essentially of a core of RNA or DNA surrounded by a protein coat.
- Either DNA or RNA, not both
- Attach, penetry, inject, replication, assembly, release
- Replication involves disassembly and reassembly
Tumors
- Tumor: An abnormal benign or malignant new growth of tissue that possesses no physiological function and arises from uncontrolled usually rapid cellular proliferation.
- Classification:
- Aggression behaviors
- Benign tumor
- Malignant tumor (a.k.a. Cancer)
- Location of the tumor
- Carcinoma (epithelial origin)
- Sarcoma (mesenchymal origin/connective tissues)
- Others (lymphoma, leukemia, melanoma, and etc.)
- Aggression behaviors
Oncogenic Virus (tumor virus):
A virus capable of causing transformation of cells, inducing a neoplasia in its host or causally linked to human tumors.
General criteria for oncogenic viruses
- The virus or part of its genome should be closely associated with the oncogenic disease (e.g., should be present in tumor tissues).
- The virus should persist throughout the disease.
- A prospective study should show that infection with the virus precedes disease.
- Prevention of virus infection (e.g., by vaccine) should prevent disease.
Why has the study of oncogenic viruses been important?
- It’s estimated that ~20% of human cancers in the world are caused by infection, of which 10-15% are caused by oncogenic viruses.
- In the past, research on oncogenic viruses led to the discovery of key cellular tumor suppressor proteins (e.g. p53) and general pathways involved in tumor development.
Studies of SV40 led to the discovery of tumor suppressor p53
DIscovery of p53
AgT → 53kDa protein interacted with AgT, which functoin as tumor suppressor.
Why has the study of oncogenic viruses been important?
- It’s estimated that ~20% of human cancers in the world are caused by infection, of which 10-15% are caused by oncogenic viruses.
- In the past, research in oncogenic viruses led to the discovery of key cellular tumor suppressor proteins (e.g. p53) and general pathways involved in tumor development.
- It can help us to identify potential diagnostic/prognostic markers and therapeutic targets for virus-mediated tumors.
Human oncogenic viruses
DNA tumor viruses:
- Small DNA tumor viruses
- Human papillomavirus (HPV)
- Simian virus 40 (SV40)
- Merkel cell polyomavirus (MCV)
- Adenovirus
- Complex DNA tumor viruses
- Epstein-Barr virus (EBV)
- Kaposi’s sarcoma-associated herpesvirus (KSHV)
- Hepatitis B virus (HBV)
RNA tumor viruses:
- Oncogenic retroviruses
- Human T-cell lymphotropic virus-1 (HTLV-1)
- Human T-cell lymphotropic virus-2 (HTLV-2)
- non-retroviruses
- Hepatitis C virus (HCV)
Why and how do oncogenic viruses cause tumors
Genetic basis of cancer
Loss of growth control:
- Genes (i.e. oncogenes) that are stimulatory for growth are hyperactive.
- Genes (i.e. tumor suppressor genes) that inhibit cell growth are turned off.
Generally, oncogenic viruses either carry a copy of oncogenes or can alter expression of the cell’s copy of these genes.
Why do oncogenic viruses want to disrupt growth control?
- It’s viruses’ way to fight against the host defense system and to meet their needs for replication and continuation of their species.
- Oncogenic viruses might accidentally cause tumors, since killing your host is a poor way to ensure your survival even if he/she is trying to kill you.
Human oncogenic viruses
DNA tumor viruses:
- Small DNA tumor viruses:
- Human papillomavirus (HPV)
- Simian virus 40 (SV40)
- Merkel cell polyomavirus (MCV)
- Adenovirus
Small DNA tumor viruses need to hijack the host DNA, RNA, and Protein synthesis systems.
- Complex DNA tumor viruses
- Epstein-Barr virus (EBV)
- Kaposi’s sarcoma-associated herpesvirus (KSHV)
- Hepatitis B virus (HBV)
Human papillomavirus (HPV)
-
More than 150 subtypes identified based on the genetic sequence of viral L1; can infect skin (~60 types) or mucosal surfaces (~40 types).
-
HPV is the most common STI in U.S.
-
16 High-risk HPVs (HR-HPV) are detected in 99% of cervical cancers. HPV- 16 and -18 cause >70% of cervical cancer cases.
-
Associated with benign papillomas (warts) and carcinomas including cervival, vulvar, vaginal, anal, penile, and head & neck squamous cell carcinoma.
-
Vaccines: HPV4 (Gardasil) targets HPV6,11,16,18) for females/males. HPV2 (Cervarix) targets HPV16,18 for females.
-
Circular, double-strand DNA virus, ~8Kb genome.
-
Viral genes:
- E1, E2: early regulatory protein
- E5, E6, E7: early oncoprotein
- L1, L2: late structural capsid protein
Human papillomavirus life cycle
Effect Skin and …
Four layer of the skin, baskm suprbasalm granular, cornifierd.
HPV first infect the basal layer and then, transferred to other layer.
DIfferenct layers express HPV gene differently because the transcription factors are different from layer to layer
Human papillomavirus (HPV) Integration and abortive replication
Abortive replication: when a virus infects a cell (or host), but cannot complete the full replication cycle, i.e. a non-productive infection.
Some gene was deleted and the replication aborted.
Mode of transformation: HPV E6 and E7
StepA: E7 prohebite RB, which free E2F1 and causing the proliferation
StepB: E7 bind p27 which regulate the CDK2/CyclineE
StepC: E7 bind to p21
Simian virus 40 (SV40)
- SV40 was first isolated as a contaminating virus in rhesus macaque monkey cells used to grow the early versions of the polio vaccine developed in the late 1950s.
- 30 millions of vaccine recipients in U.S. accidentally received vaccine doses with low to high titers of the SV40 between 1955 and 1963.
- SV40 can induce tumors including primary brain cancers, malignant mesotheliomas, bone tumors, and systemic lymphomas in test animals.
Simian virus 40 in human malignant disease
- In 2002, the National Academy of Sciences Immunization Safety Review committee that stated, "The committee concludes that the biological evidence is moderate that SV40 exposure could lead to cancer in humans under natural conditions.”
- In 2004, the National Cancer Institute announced that “substantial epidemiological evidence has accumulated to indicate that SV40 likely does not cause cancer in humans”.
- In 2006, new evidence indicated that SV40 is involved in the development of peritoneal and pleural mesothelioma in human.
Simian virus 40 (SV40)
- Belongs to polyomavirus family.
- Nonenveloped, circular dsDNA genome, 5kb in size.
- Genome is divided into early and late regions.
- Oncogene: Large T (tumor) antigen.
SV40 large T-antigen
TAg bind to the pRb → Rb degnation
TAg bind to p53
Paper Discussion Merkel cell polyomavirus (MCV)
- Feng H, Shuda M, Chang Y, Moore PS. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science. 2008 Feb 22;319(5866):1096-100. PubMed PMID: 18202256.
- Shuda M, Feng H, Kwun HJ, Rosen ST, Gjoerup O, Moore PS, Chang Y. T antigen mutations are a human tumor-specific signature for Merkel cell polyomavirus. Proc Natl Acad Sci U S A. 2008 Oct 21;105(42):16272-7. PubMed PMID: 18812503.
Adenovirus
- Linear, non-segmented dsDNA genome.
- 7 groups, 57 serotypes in humans.
- Associated with respiratory disease, conjunctivitis, and gastroenteritis.
- Certain adenovirus subtypes can transform mammalian cells (e.g. adenovirus group c serotype 5).
Adenovirus mediated oncogenesis
- Oncogenes:
- E1A
- E1B
Oncogenesis mediated by small DNA tumor virus
- In benign lesions induced by the small DNA tumor viruses, viral genomes are typically maintained extrachromosomally. Malignant progression is often associated with viral integration into host cell chromatin.
- Integration usually leads to abnormal expression of early genes.
- Early genes promote cell cycle progression and prevent apoptosis.
- HPV - E7 (cell cycle) and E6 (apoptosis)
- SV40 - T Ag (cell cycle and apoptosis)
- Adenovirus - E1A (cell cycle) and E1B (apoptosis)
Human oncogenic viruses
- DNA tumor viruses: Complex DNA tumor viruses
- Epstein-Barr virus (EBV)
- Kaposi’s sarcoma-associated herpesvirus (KSHV)
- Hepatitis B virus (HBV)
Epstein-Barr virus (EBV)
- EBV belongs to human herpes virus family (a.k.a. HHV-4).
- Infect >90% of the adult population in U.S (EveryBody’s Virus).
- Infect lymphocytes and epithelial cells.
- Transmitted by saliva.
- Generally, don’t need to integrate.
- Cause lymphoma in Marmosets and transform human B-lymphocytes in vitro.
EBV in human diseases
- Malignant diseases:
- Burkitt’s lymphoma in the tropics, where it is more common in malaria-endemic regions.
- Nasopharyngeal carcinoma, particularly in China and southeast Asia, where certain diets may act as co-carcinogens.
- B cell lymphomas in immune suppressed individuals (such as in organ transplantation or HIV).
- Hodgkin’s lymphoma in which it has been detected in a high percentage of cases (about 40% of affected patients).
- Non-Malignant diseases:
- Infectious mononucleosis (a.k.a. kissing disease, glandular fever)
- Autoimmune diseases including dermatomyositis, systemic lupus erythematosus, rheumatoid arthritis, and multiple sclerosis.
Epstein-Barr Virus (EBV)
- Enveloped, linear dsDNA genome in virions; circular dsDNA in host cells.
- ~171kb genome. Latency genes and lytic genes.
EBV life cycle
- There are 2 stages in EBV’s life cycle: latency and lytic replication (reactivation).
- Latency: viral persistence.
- Lytic replication: production of new infectious virions.
- EBV Encodes its own polymerases and don’t need S-phase environment for replication.
EBV latency programs
EBV latent genes
- EBNA2 and LMP1 are absolutely required for EBV-mediated transformation of B cells.
- EBNA-LP, EBNA3A and EBNA3C play a critical role in the transformation process.
EBV Nuclear Antigen 2 (EBNA2)
- First virus protein expressed after infection of B cells.
- Plays a pivotal role in B cell immortalization.
- Transcription factor, regulate the expression of viral LMP1 and LMP2 as well as cellular oncogene c-Myc.
Latent Membrane Protein 1 (LMP1)
- Classical oncogene (transgenic mice expressing LMP1 alone in B cells develop lymphomas).
- Acts as constitutively activated tumor necrosis factor receptor (TNF-R) CD40.
- Inhibits apoptosis by upregulating BCL2 and A20.
- Activates NF-kB pathway and JAK/STAT pathway.
EBV Nuclear Antigen 3C (EBNA3C)
- Essential for B-cell transformation in vitro.
- Cell cycle regulator.
Kaposi’s sarcoma-associated herpesvirus (KSHV)
- Discovered in 1994, belongs to the herpes virus family (a.k.a. HHV-8).
- ~10% of the general population has been infected in U.S.
- Transmitted through sexual contact and organ transplantation.
- Causes Kaposi’s sarcoma, body cavity-based lymphoma, and Castleman’s disease.
- Enveloped dsDNA genome.
- Latency and lytic replication.
- Molecular piracy (molecular mimicry).
- Major latent genes: LNA-1, v-cyclin, and v- FLIP.
Regulation of cell cycle by KSHV LNA-1 and v-Cyclin
LNA1 → RB; p53; Ring3
Hepatitis B Virus (HBV)
- Belongs to Hepadnavirus family.
- Circular Partially dsDNA genome.
- Causes hepatitis B, cirrhosis, and hepatocellular carcinoma.
HBV mediated oncogenesis
Mechanism is not clear.
- Integration disrupts oncogene or tumor- suppressor genes.
- Hepatitis B virus X (HBx) protein.
Targeted therapies for EBV-associated malignancies
T-cell-based adoptive immunotherapeutics for EBV-associated malignancies
16 Nuclear Receptors|Tulane
https://karobben.github.io/2021/11/30/LearnNotes/tulane-cellbio-16/